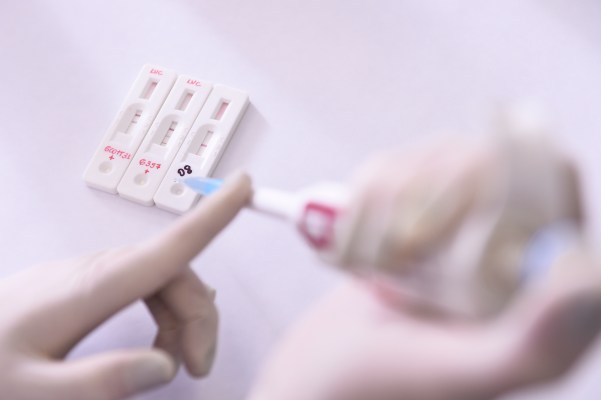
The FDA just okayed multiple 15-minute blood tests to screen for coronavirus, but there are caveats
On Thursday, the FDA amended their emergency policy around diagnostic testing for SARS-CoV-2, the novel coronavirus that causes COVID-19. Following on a change made March 16, the agency opened the door for a number of specific private entities and labs to develop and distribute tests that can provide results on the spot in as little […]
TechCrunch - Click to read the rest of the news
Önemli Not: Bu haberlerin hukuki muhatabı haber kaynaklarıdır. Haberlerle ilgili her tür şikayetinizi adresimize gönderebilirsiniz.